Difference between osmosis and diffusion often becomes clear during a simple science experiment. Imagine placing a drop of ink in water it spreads slowly on its own. Now think of plant roots absorbing water. Both processes involve movement, but they are not the same.
The difference between osmosis and diffusion lies in how substances move and what kind of particles are involved. Both are natural processes that do not need energy, yet they follow different rules.
Many students confuse them because both involve movement from high concentration to low concentration. However, understanding the difference between osmosis and diffusion helps you grasp important biology concepts.
Let’s explore them step by step.
Key Difference Between the Both ⚖️
The main difference between osmosis and diffusion is:
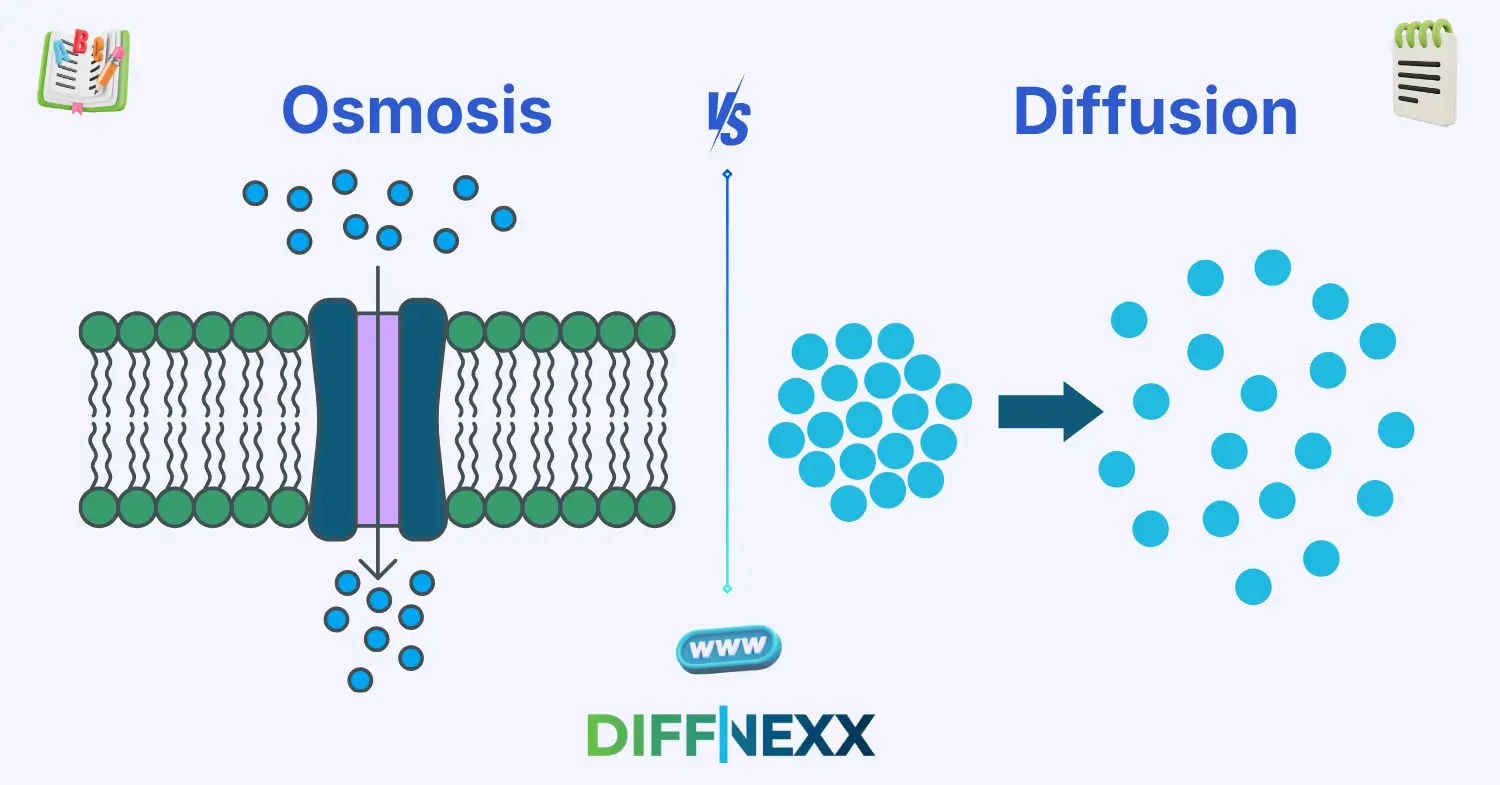
- Osmosis involves movement of water only through a semi-permeable membrane.
- Diffusion involves movement of any particles (like gases or liquids) without needing a membrane.
This key idea explains most of their differences.
Importance – Why Knowing the Difference Matters 🎯
Understanding the difference between osmosis and diffusion is essential for learners and professionals.
For students, it builds a strong foundation in biology and chemistry. It helps in exams and practical experiments.
For professionals like doctors and scientists, these concepts explain how cells work and how substances move in the body.
In society, this knowledge helps us understand everyday processes like plant growth, food preservation, and even breathing.
Pronunciation Section 🔊
Osmosis
US: /ɒzˈmoʊ.sɪs/
UK: /ɒzˈməʊ.sɪs/
Diffusion
US: /dɪˈfjuː.ʒən/
UK: /dɪˈfjuː.ʒən/
Now that you can pronounce them correctly, let’s define them simply.
Core Definitions 📚
Osmosis
Osmosis is the movement of water across a semi-permeable membrane from an area of high water concentration to low water concentration. It is essential for maintaining cell balance.
Example: Plant roots absorb water from the soil through osmosis.
Diffusion
Diffusion is the movement of particles (like gases or liquids) from a region of high concentration to low concentration. It happens naturally without energy.
Example: Perfume smell spreading in a room is diffusion.
10 Clear Differences Between Osmosis and Diffusion 🔬
1. Type of Substance
Osmosis involves only water. Diffusion involves any particles.
- Example for Osmosis: Water moves into a cell.
- Example for Diffusion: Oxygen spreads in air.
2. Membrane Requirement
Osmosis requires a semi-permeable membrane. Diffusion does not always need one.
- Example for Osmosis: Occurs across cell membranes.
- Example for Diffusion: Occurs freely in air.
3. Medium
Osmosis happens in liquids. Diffusion occurs in solids, liquids, and gases.
- Example for Osmosis: Water in plant cells.
- Example for Diffusion: Gas spreading in air.
4. Function
Osmosis maintains water balance. Diffusion spreads substances evenly.
- Example for Osmosis: Keeps cells hydrated.
- Example for Diffusion: Distributes nutrients.
5. Direction of Movement
Both move from high to low concentration, but osmosis focuses on water balance.
- Example for Osmosis: Water enters a dry cell.
- Example for Diffusion: Smoke spreads in air.
6. Speed
Diffusion can be faster in gases. Osmosis is usually slower.
- Example for Osmosis: Gradual water movement.
- Example for Diffusion: Quick spreading of smell.
7. Energy Requirement
Both do not require energy (passive processes).
- Example for Osmosis: Happens naturally.
- Example for Diffusion: Happens naturally.
8. Role in Biology
Osmosis is vital for cell survival. Diffusion supports gas exchange.
- Example for Osmosis: Helps plant cells stay firm.
- Example for Diffusion: Helps lungs exchange gases.
9. Pressure Influence
Osmosis is affected by osmotic pressure. Diffusion depends on concentration gradient.
- Example for Osmosis: Pressure controls water flow.
- Example for Diffusion: Concentration controls spread.
10. Application
Osmosis is used in plant and cell processes. Diffusion is used in many natural and industrial processes.
- Example for Osmosis: Water absorption in roots.
- Example for Diffusion: Mixing of gases in air.
Why Knowing the Difference Matters 🎯
Students need this knowledge to understand basic science concepts and perform experiments correctly.
Professionals use it in medicine, biology, and chemistry to study cells and body systems.
In everyday life, it helps explain how nature works around us.
Real-world Consequences of Confusion
Confusing these processes can lead to misunderstanding scientific experiments or biological systems.
Why People Get Confused 🤔
Similar Concepts
Both involve movement from high to low concentration.
Scientific Terms
The names sound complex and similar.
Context Overlap
Both are taught together in science classes.
Informal Understanding
People simplify both as “movement,” ignoring differences.
Connotation & Emotional Tone 🎭
Connotation = the emotional meaning associated with a word.
Osmosis
- Positive: Balance, stability
- Neutral: Biological process
- Example: Osmosis suggests controlled movement.
Diffusion
- Positive: Spread, expansion
- Neutral: Natural process
- Example: Diffusion suggests free movement.
Usage in Metaphors, Similes & Idioms 🗣
- “Knowledge spreads like diffusion” → ideas spreading
- “Ideas flow through osmosis” → gradual learning
Examples:
- Ideas spread through the group like diffusion.
- He learned the language through osmosis.
Comparison Table 📊
| Feature | Osmosis | Diffusion |
|---|---|---|
| Meaning | Water movement | Particle movement |
| Tone | Controlled | Free |
| Usage | Biological cells | General processes |
| Context | Requires membrane | May not require membrane |
| Formality | Scientific | Scientific |
Which Is Better in What Situation? ⚖️
When to Use Osmosis
Use osmosis when discussing water movement across membranes, especially in biology. For example, in plant cells or human cells.
When to Use Diffusion
Use diffusion when talking about general movement of particles, such as gases or liquids spreading.
Situational Clarity
Choose based on whether water or general particles are involved.
Contextual Correctness
Using the right term ensures accuracy in science discussions.
Literary or Cultural References 📖
- Book: Biology by Neil A. Campbell (Science, 1987)
- Documentary: The Secret Life of Plants (USA, 1979)
These explain biological processes in detail.
FAQs ❓
1. Is osmosis a type of diffusion?
Yes, osmosis is a special type of diffusion that involves only water. It requires a semi-permeable membrane.
2. Do both require energy?
No, both are passive processes. They happen naturally without using energy.
3. Where does osmosis occur?
Osmosis mainly occurs in living cells, especially in plants and animals.
4. Can diffusion happen in gases?
Yes, diffusion is very common in gases. For example, air spreads in a room.
5. How can I remember the difference?
Think: Osmosis = water + membrane. Diffusion = any particles + no barrier needed.
Conclusion 🏁
The difference between osmosis and diffusion is simple but important. Osmosis involves the movement of water through a membrane, while diffusion involves the movement of any particles without needing a barrier.
Understanding this difference helps you learn biology more effectively and apply it in real life. It also improves your scientific thinking and clarity.
Keep exploring science concepts like these they make the world easier to understand and more interesting.

Eliza Marlowe is a passionate content strategist and comparison writer at diffnexx.com. She specializes in breaking down complex “difference between” topics into clear, reader-friendly guides. With a background in linguistics and digital research, Eliza focuses on accuracy, clarity, and content that helps readers make informed decisions with confidence.