Difference between element and compound often comes up in science class. Imagine you are in a lab, holding a piece of gold and a glass of water. Both are substances but are they the same type? Not quite.
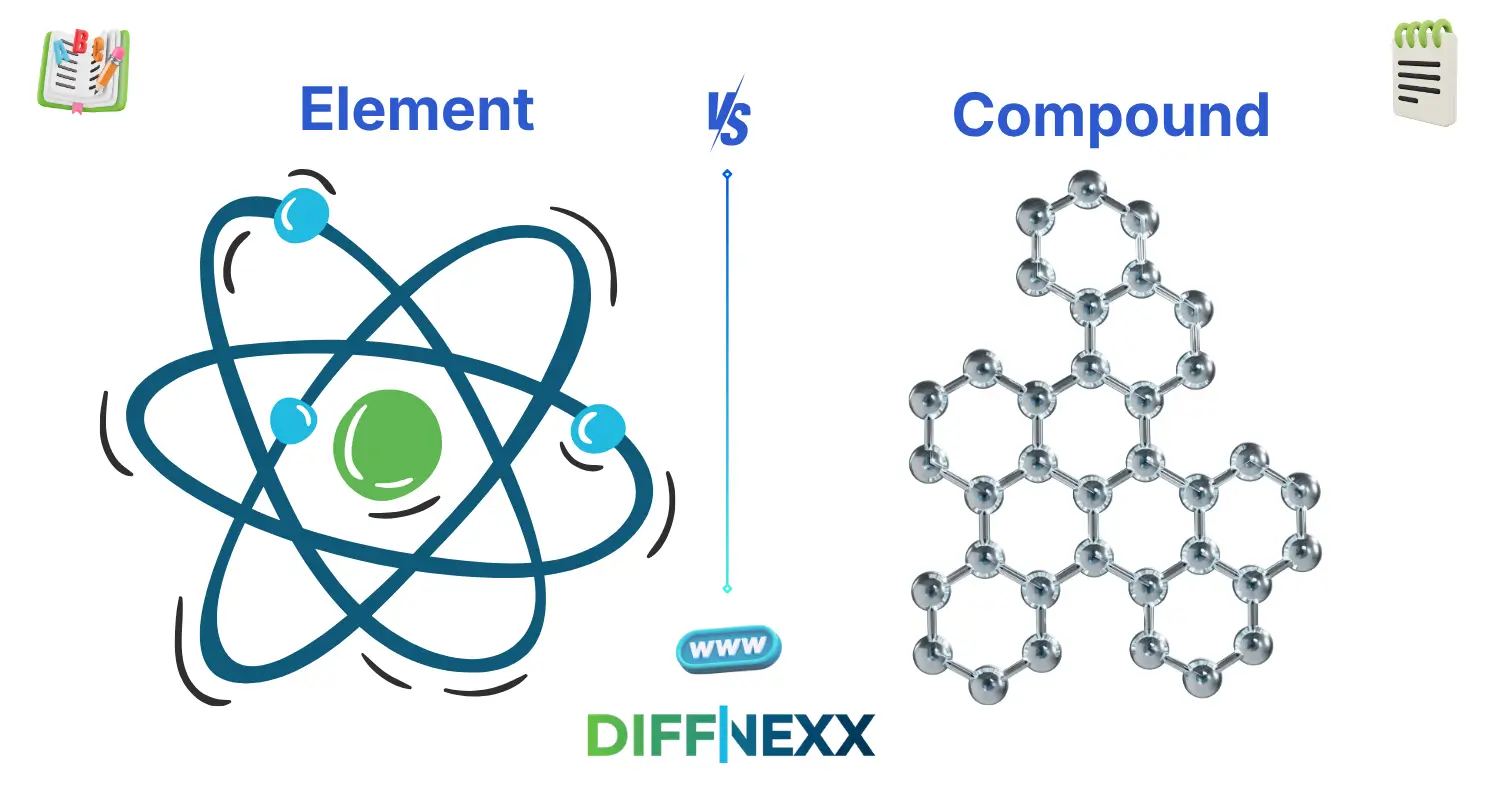
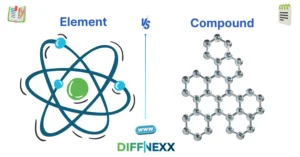
The way substances are made determines the difference between element and compound. An element is a pure substance that consists of only one kind of atom. A compound, on the other hand, contains two or more different atoms that chemically combine.
By understanding the difference between element and compound, you can see how matter is built. It also explains why water behaves differently from oxygen or iron.
Let’s break it down step by step so everything becomes clear.
Key Difference Between the Both ⚖️
The main difference is simple:
- An element contains only one type of atom.
- A compound contains two or more different atoms chemically bonded.
Elements are basic building blocks. Compounds are combinations of those blocks.
Importance – Why Their Difference Matters 🌍
Knowing the difference between element and compound is essential in science and daily life.
For students, it builds a strong foundation in chemistry. It helps you understand reactions, formulas, and the structure of matter.
For professionals like chemists and engineers, this knowledge is critical. It guides how materials are created, tested, and used safely.
In society, it supports industries like medicine, agriculture, and manufacturing. Understanding substances helps improve products and protect the environment.
🔊 Pronunciation Section
Element
US: /ˈelɪmənt/
UK: /ˈelɪmənt/
Compound
US: /ˈkɑːmpaʊnd/
UK: /ˈkɒmpaʊnd/
Now that you know how to say them, let’s explore what they mean.
📚 Core Definitions
Element
An element contains only one type of atom. You can not break it into simpler substances by chemical means.
- Example: Gold (Au) is an element made only of gold atoms.
Compound
A compound consists of two or more different elements chemically bonded together. It has a fixed ratio of atoms.
- Example: Water (H₂O) is a compound made of hydrogen and oxygen.
📚 10 Clear Differences Between Element and Compound
1. Basic Nature
An element is a single type of atom. A compound is a combination of different atoms.
- Example for Element: “Oxygen is an element.”
- Example for Compound: “Carbon dioxide is a compound.”
2. Composition
Elements have one kind of atom. Compounds have two or more kinds.
- Example for Element: “Iron contains only iron atoms.”
- Example for Compound: “Salt contains sodium and chlorine.”
3. Chemical Bonding
Elements do not need bonding with other types. Compounds require chemical bonds.
- Example for Element: “Helium exists alone.”
- Example for Compound: “Water forms through bonding.”
4. Breakdown Ability
You cannot break Elements chemically, while you can break compounds into elements.
- Example for Element: “You cannot break gold into simpler substances.”
- Example for Compound: “Water can split into hydrogen and oxygen.”
5. Representation
Elements use symbols. Compounds use chemical formulas.
- Example for Element: “O represents oxygen.”
- Example for Compound: “H₂O represents water.”
6. Properties
Elements have unique properties. Compounds have new properties different from their elements.
- Example for Element: “Oxygen supports burning.”
- Example for Compound: “Water puts out fire.”
7. Types
The periodic table includes a limited number of elements. Compounds are almost unlimited.
- Example for Element: “There are about 118 elements.”
- Example for Compound: “Millions of compounds exist.”
8. Physical State
Elements can be solid, liquid, or gas. Compounds can also exist in all states but differ widely.
- Example for Element: “Mercury is liquid.”
- Example for Compound: “Water is liquid at room temperature.”
9. Formation Process
Elements form naturally. Compounds form through chemical reactions.
- Example for Element: “Gold is found in nature.”
- Example for Compound: “Rust forms from iron and oxygen.”
10. Stability
Elements are stable in their form. Compounds depend on bonds for stability.
- Example for Element: “Noble gases are stable.”
- Example for Compound: “Compounds break under certain conditions.”
🎯 Why Knowing the Difference Matters
Students need this knowledge to understand chemistry basics. It helps in exams and builds logical thinking.
Professionals use this distinction in labs and industries. It ensures correct handling of materials and safe reactions.
In daily life, it improves awareness of what we use. From water to medicines, everything depends on compounds and elements.
Real-world Consequences of Confusion
Confusing them can lead to mistakes in experiments. It can also cause misunderstanding in education and communication.
🧠 Why People Get Confused
Similar Concepts
Both are types of substances, so they seem alike.
Semantic Overlap
They are closely related in chemistry.
Context-Based Usage
People often use them without precision.
Informal Speech
Everyday language ignores scientific accuracy.
🎭 Connotation & Emotional Tone
Connotation = the emotional meaning associated with a word.
Element
Neutral and scientific. It feels basic and foundational.
- Example: “Hydrogen is the simplest element.”
Compound
Also neutral but slightly more complex.
- Example: “This compound has a strong reaction.”
🗣 Usage in Metaphors, Similes & Idioms
People sometimes use these terms metaphorically:
- “Trust is an element of success.”
- “Teamwork is a compound of skills.”
Examples:
- “Honesty is a key element of leadership.”
- “Success is a compound of effort and patience.”
📊 Comparison Table
| Feature | Element | Compound |
|---|---|---|
| Meaning | Single type of atom | Two or more atoms combined |
| Tone | Basic, scientific | Slightly complex |
| Usage | Chemistry basics | Chemical combinations |
| Context | Scientific study | Reactions and materials |
| Formality | Formal | Formal |
⚖️ Which Is Better in What Situation?
When to Use Element
Use “element” when talking about basic substances. It fits well in science lessons and simple explanations.
When to Use Compound
Use “compound” when describing substances made from different elements. It is common in chemistry discussions.
Situational Clarity
Always identify whether the substance is pure or combined. This helps you choose the right term.
Contextual Correctness
In scientific writing, accuracy matters. Using the correct term avoids confusion and improves clarity.
📖 Literary or Cultural References
- Book: The Disappearing Spoon (Science, Sam Kean, 2010) – Explores elements and their stories.
- Movie: Flubber (USA, 1997) – Shows fictional compounds and chemical creativity.
❓ FAQs
1. Can an element become a compound?
Yes, when elements react chemically, they form compounds. For example, hydrogen and oxygen combine to form water.
2. Are all substances either elements or compounds?
No, mixtures also exist. They are different because their components are not chemically bonded.
3. Why can compounds be broken down but not elements?
Compounds form through bonds that can be broken by chemical reactions. Elements are already in their simplest form.
4. Is air an element or a compound?
Air is a mixture, not a compound. It contains gases like nitrogen and oxygen.
5. How many elements are there?
Scientists have identified about 118 elements. They continue to study and discover more.
🏁 Conclusion
The difference between element and compound is fundamental to understanding chemistry. Elements are pure substances made of one type of atom, while compounds are formed by combining different atoms in fixed ratios.
This distinction helps explain how matter behaves and changes. It also supports learning, research, and practical applications in everyday life.
By understanding these terms, you build a strong scientific foundation. Keep exploring, stay curious, and let science guide your thinking forward 🔬✨.

Anton Reyes is a contributing writer at DiffNexx.com, where he explores meaningful differences in language, culture, and everyday concepts. With a background in creative nonfiction, Anton brings clarity and balance to complex comparisons. His writing helps readers understand subtle distinctions in words and ideas, making learning both insightful and engaging.